How Biologics and Biosimilars Work
Though the exact cause of chronic inflammatory conditions like psoriasis is unknown, dynamic factors and processes that drive inflammation include over-activated immune cells and cell signalling chemicals called cytokines . Biologic and biosimilar drugs (going forward referred to as biologic agents) target specific cytokines to suppress their overproduction and to bring the immune system back to normal (see figure below).
Cytokines that are known to be involved in inflammation in psoriasis and psoriatic arthritis include tumour necrosis factor, interleukin 17, interleukin 12 and interleukin 23. Each of these molecules are specific messengers in the immune system involved in the development of psoriasis.
TNF (Tumour necrosis factor (often called tumor necrosis factor alpha or TNF))
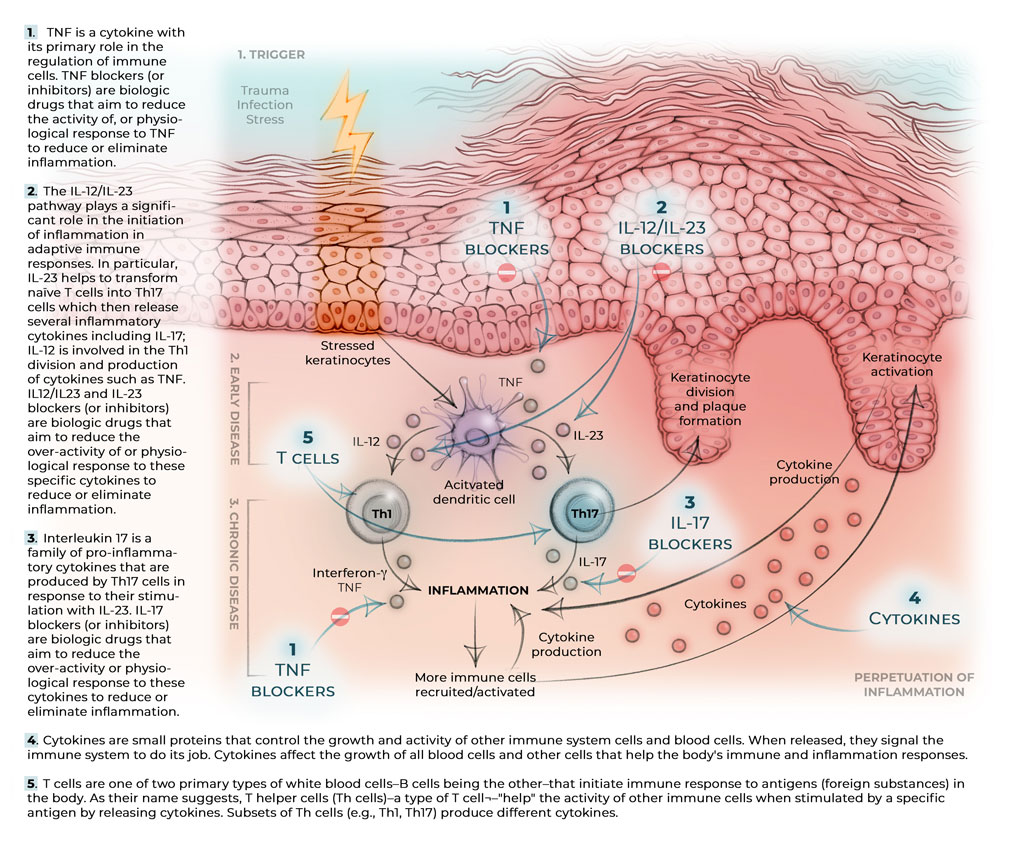
TNF is a cytokine with its primary role in the regulation of immune cells. When immune cells detect an infection, they release TNF to alert other immune system cells as part of an inflammatory response. A TNF blocker (or inhibitor) is a biologic drug that aims to reduce the activity of, or physiological response to, TNF to reduce or eliminate inflammation. The TNF blockers used for moderate-to-severe forms of plaque psoriasis and psoriatic arthritis are adalimumab, etanercept, infliximab and certolizumab pegol. Golimumab is a TNF blocker indicated to treat moderate-to-severe psoriatic arthritis. TNF blockers are broad-acting agents, as TNF is an upstream mediator of inflammation, meaning that it comes earlier in the inflammatory signalling process for psoriatic disease. The dosing regimen, side effect profiles and other special considerations may differ, depending on the drug.
IL-12/IL-23 (Interleukin 12(IL-12) and Interleukin 23 (IL-23))
Interleukins (ILs) are a group of cytokines that are released by white blood cells.
The IL-12/IL-23 pathway plays a significant role in the initiation of inflammation in adaptive immune responses. In particular, IL-23 helps to transform naïve T helper cells into Th17 cells which then release several inflammatory cytokines including IL-17; IL-12 helps with the Th1 division and production of cytokines such as interferon-γ and tumour necrosis factor (TNF). The dosing regimen, side effect profiles and other special considerations may differ, depending on the drug.
The only IL-12/23 blocker available is ustekinumab. It is used for moderate-to-severe forms of plaque psoriasis and psoriatic arthritis.
The IL-23 blockers are the newest class of biologics in the treatment of psoriasis. They include risankizumab, guselkumab, and tildrakizumab which are all available in Canada to treat moderate-to-severe plaque psoriasis and, in the case of guselkumab, psoriatic arthritis.
The dosing regimen, side effect profiles and other special considerations may differ, depending on the drug.
IL-17 (Interleukin 17)
Interleukins (ILs) are a group of cytokines that are released by white blood cells.
Interleukin 17 is a family of pro-inflammatory cytokines that are produced by a type of T helper cells (a type of white blood cell) known as T helper 17 cell in response to their stimulation with IL-23. IL-17 blockers (or inhibitors) include secukinumab, ixekizumab, and brodalumab and they treat moderate-to-severe forms of plaque psoriasis and psoriatic arthritis. IL-17 cytokines are important for the immune function of barrier tissues including the skin, the lungs and the gastrointestinal tract. Patients with active chronic inflammatory bowel disease (e.g., Crohn’s disease, ulcerative colitis) should not receive IL-17 blockers because of potential aggravation of these conditions. The dosing regimen, side effect profiles and other special considerations may differ, depending on the drug.
Abatacept
Abatacept is used to treat psoriatic arthritis. It does not block cytokines like TNF but rather attaches to the surface of inflammatory cells and blocks communication between these cells. By blocking this communication, abatacept lessens inflammation. Though it can be a first-line treatment, abatacept is often prescribed when patients with moderate to severe psoriatic arthritis who have not responded to one or more disease-modifying antirheumatic drugs (DMARDs), such as methotrexate, or other biologic drugs.
Since many factors are involved in chronic inflammation, what drives this process in one individual may differ from another individual with the same chronic illness or at different stages of disease. A single biologic agent that resolves inflammation in one patient may not work to the same degree as another patient with the same disease or may lose its efficacy for an individual over time.
Moreover, many factors go into making treatment decisions – including topicals, phototherapy, systemic drugs and biologic agents – throughout a patient’s life. Factors to consider include insurance coverage, mode of delivery (systemic versus topical; intravenous injection versus subcutaneous injection versus oral), dosing profile, family planning and/or potential for pregnancy, medical history and comorbidities.
Treatment factors to consider
At this stage, since we do not have biomarkers that can predict which drug will work best in an individual patient with psoriasis or psoriatic arthritis, there is a certain level of trial and error to find the right treatment for the right individual over the course of their condition. Individuals may respond to one biologic agent, and not another even within the same class (e.g., TNF blockers; IL-17 blockers etc.) and/or treatment can work for a certain amount of time and then lose its efficacy in an individual.
Many factors go into deciding which treatment – including topicals, phototherapy, systemic drugs and biologic agents – is right for the individual throughout a patient’s life. Factors to consider include insurance coverage, mode of delivery (systemic versus topical; intravenous injection versus subcutaneous injection versus oral), dosing profile, family planning and/or potential for pregnancy, medical history and comorbidities.
Moreover, some biologics may be preferred over others for certain types/locations of psoriasis (such as scalp psoriasis, nail psoriasis, genital psoriasis and palmoplantar disease).
Ongoing shared decision making between a patient and their dermatologist/rheumatologist/healthcare team is crucial in ensuring that a person gets the care, treatment and support that they need to live well with psoriatic disease.
References
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7669769/
- https://www.canada.ca/content/dam/hc-sc/migration/hc-sc/dhp-mps/alt_formats/pdf/brgtherap/applic-demande/guides/Fact-Sheet-EN-2019-08-23.pdf
- https://www.canada.ca/en/health-canada/services/drugs-health-products/biologics-radiopharmaceuticals-genetic-therapies/biosimilar-biologic-drugs.html
- https://www.canada.ca/en/health-canada/services/health-care-system/pharmaceuticals/access-insurance-coverage-prescription-medicines/provincial-territorial-public-drug-benefit-programs.html
- https://www.canada.ca/en/health-canada/services/health-care-system/pharmaceuticals/access-insurance-coverage-prescription-medicines/federal-public-drug-benefit-programs.html
- https://www.cadth.ca/sites/default/files/symp-2018/presentations/april16-2018/Concurrent-Session-A5-Mary-Lou-Robertson.pdfhttps://www2.gnb.ca/content/dam/gnb/Departments/h-s/pdf/en/NBDrugPlan/biosimilars/Patient-Support-Programs_Biosimilars-Initiative.pdf
Last updated October, 2022
